Performed by Dr. Apurva Patel (with his team), Hip Preservation & Replacement Surgeon in Ahmedabad
Patient’s Problem
Ms. Dhrasti(Name Changed for Privacy), a 22-year-old woman with known Sickle Cell Disease (SCD), visited our clinic with persistent and severe right-hip pain lasting for several months. The pain significantly restricted her ability to walk, stand, or perform daily activities. She feared losing mobility at a young age and was seeking a safe treatment that could prevent the condition from progressing.
Diagnosis
Clinical evaluation, X-rays, and MRI revealed:
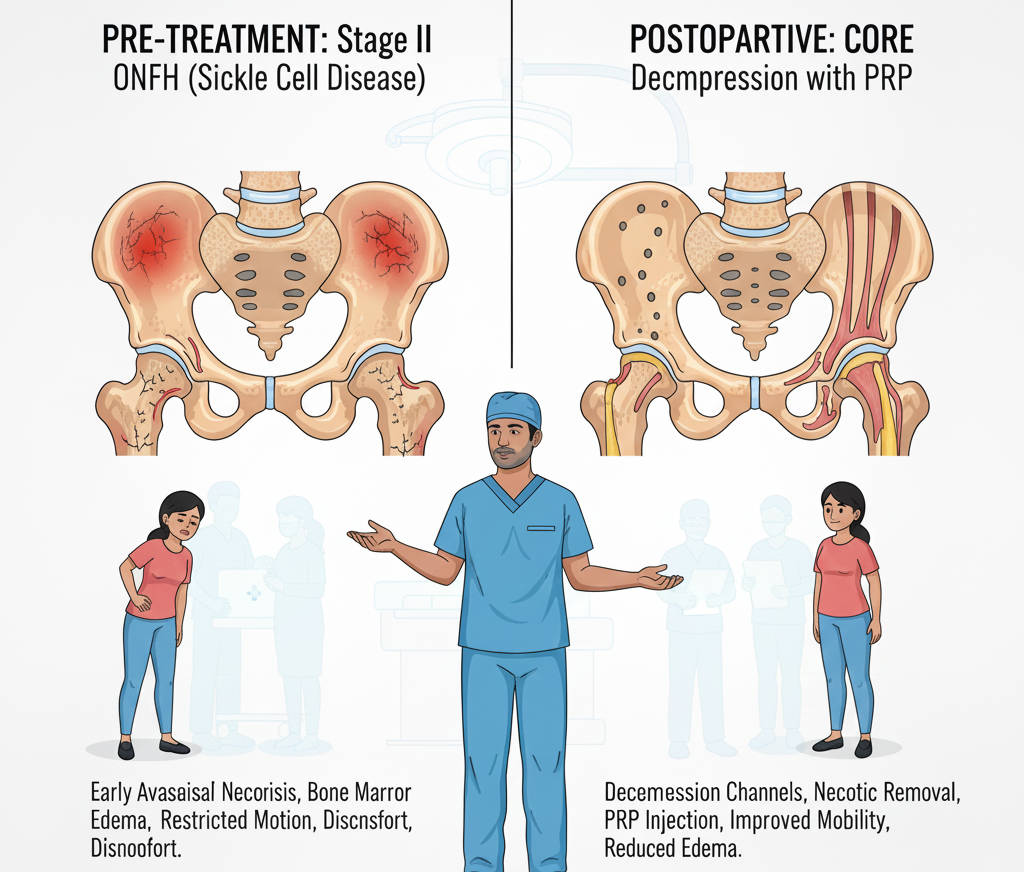
- Osteonecrosis of the Femoral Head (ONFH)
- Stage II disease according to the Modified Ficat & Arlet classification
- Bone marrow edema
- Restricted hip movements
- Pain affecting daily activities
Because the femoral head had not yet collapsed, timely intervention was essential to preserve the hip joint and avoid future total hip replacement.
Treatment Approach
The treatment plan followed the same protocol described in the 2023 research article:
“Outcomes of Core Decompression with Platelet Rich Plasma and Oral Ibandronate Supplementation in Osteonecrosis of the Femoral Head in Sickle Cell Positive Young Adults”
Medical Team & Their Roles
The treatment and management were performed according to the protocol designed and executed by the following specialists from PIMSR, Parul University, Vadodara:
- Dr. Arvind Kumar – Professor, Department of Orthopaedics
- Lead surgeon and primary designer of the treatment protocol.
- Dr. Apurva Patel – Senior Resident, Orthopaedics
- Assisted in surgical planning and operative execution.
- Dr. Sonal Lakum – Assistant Professor, Microbiology
- Responsible for timely PRP preparation and microbiological safety.
- Dr. Mayank Gajera – Junior Resident, Orthopaedics
- Assisted intra-operatively and in postoperative patient monitoring.
- Dr. Harshil Barot – Junior Resident, Orthopaedics
- Assisted the surgical team and rehabilitation planning.
- Dr. Aliasgar Rampurwala – Assistant Professor, Orthopaedics
- Supported surgical decisions, postoperative evaluation, and follow-up assessment.
This exact team performed the protocol described in the research and contributed to the outcomes and methodology.
Surgical Procedure: Core Decompression + PRP + Oral Ibandronate
The surgery followed the evidence-based technique described in the research paper:
1. Core Decompression
- Two 6 mm decompression channels were created using a hollow mill under C-arm guidance.
- Necrotic (dead) bone was evacuated.
- Any depressed articular surface was elevated when needed.
2. Platelet-Rich Plasma (PRP) Injection
- 15 ml of blood was collected and processed by the microbiology lab.
- 4 ml of fresh PRP was prepared immediately and injected into the decompressed bone tracts.
- Both channels were packed with bone wax for stability.
3. Oral Ibandronate Therapy
- Ibandronate 150 mg once monthly, started before surgery and continued for 24 weeks.
- Helps suppress bone resorption and prevent early collapse.
4. Physiotherapy Protocol
- Day 1: Quadriceps and ankle pump exercises
- Day 2: Hip–knee–ankle range of motion
- Partial weight-bearing: 6th week
- Full weight-bearing: 12th week
Results: Before & After Transformation
Before Treatment
- Severe hip pain
- Limited walking ability
- Restricted hip movement
- Daily activity limitation
- Fear of disability
After Treatment
Based on similar Stage II cases in the study and Ms. Dhrasti’s follow-up:
- Significant pain reduction immediately after surgery
- Improved hip function and activity levels
- Better range of motion
- MRI showed a reduction of bone marrow edema and improved vascularity
- No early postoperative complications
- Ability to gradually resume a normal lifestyle
- Functional improvement aligned with research outcomes, where:
- 80% of Stage I/II patients achieved excellent to good results
- Mean Modified Harris Hip Score improved significantly
Ms. Dhrasti’s results matched the research findings, indicating good-to-excellent recovery.
Conclusion
This case study, aligned with the 2023 research article, demonstrates that Core Decompression combined with PRP and Oral Ibandronate is:
- Safe
- Minimally invasive
- Cost-effective
- Highly effective in Stage I and Stage II ONFH in Sickle Cell Disease
The coordinated expertise of:
- Dr. Arvind Kumar,
- Dr. Apurva Patel,
- Dr. Sonal Lakum,
- Dr. Mayank Gajera,
- Dr. Harshil Barot,
- Dr. Aliasgar Rampurwala
ensured precise execution of the protocol and successful patient outcomes.
With timely intervention and evidence-based practice, young SCD patients Ms. Dhrasti, can avoid early femoral head collapse and return to pain-free, active living.